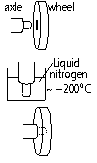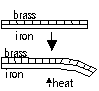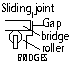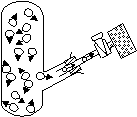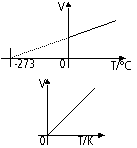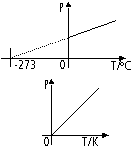Phys9
THERMAL PHYSICS
KINETIC THEORY OF MATTER
Kinetic theory- attempts to explain how small scale interactions of atoms or molecules can combine to cause large scale behaviour
Kinetic Molecular Model of Matter
-a model can be designed to explain properties of states of matter
-assumptions; 1matter made up of particles (atoms) / molecules- groups of joined atoms & 2particles in continuous random motion
-a molecule of a substance =smallest particle of a substance which can exist under normal circumstances & retain all properties of subs
-general thickness of a molecule ~ 10-9 - ~10-10m
-every substance can exist in any of the three states under right conditions of temperature and pressure
-according to kinetic theory atoms or molecules that compose matter in a particular state are held together by intermolecular / inter-atomic forces (electrical forces, both attractive and repulsive that molecular exert on each other when the are close together)
Solids
-regular, geometrical structure
-molecules closely together; regular pattern = space- lattice > high density
-incompressible
-fixed vol + shape
-typical interatomic dist in a crystal is ~ 3 x 10-10 m
strong intermolecular attractive &repulsive forces > 1molecule motion limited to vibrations about fixed pos + 2fixed vol & shape
temp ^: KE of vibrations ^ > vibrations larger >separation bet molecules slightly ^
mp: molecules vibrate violently > collide, attractive forces cant hold molecules together > space lattice collapses > solid melts / liquefies, work required to overcome forces = latent heat of fusion
Liquids
-molecules slightly further apart than in solid > high density
-fixed vol but not shape > take shape of container and can flow
-incompressible
-order of separation of molecules about same for solids
occur in clusters > tendency to form droplets + high density
intermolecular forces are less strong than in solid > molecules vibrate & move rapidly, randomly, continuously throughout liquid
random motion throughout liquid, average kinetic energy of molecules increases w/ temp, presence of attractive, cohesive forces bet molecules > 1fixed vol + 2pulling back of molecules near surface of liquid, opposing escape, more energetic (more kinetic + heat energy) ones can escape > evaporation > cooling (heat taken away)
Gases
-molecules very far apart > low density
-no fixed vol or shape > take up any space available
-compressible
-at stp (standard temp & pressure), av.separation of gas molecules ~ 33 x 10-10 m (10x further apart than in solids & liquids) (density of gas ~1/1000 density of liquid or solid, ρ gas ~ 1/1000 ρ liquid)
intermolecular forces are very weak (~0) > random movement at very high speeds (~500m/s), av KE ^ proportionately w/ temp, negligible attractive / repulsive forces bet atoms as they are far apart > no fixed vol / shape,
Temperature
Temperature is looked on as a rough measure of KE of the molecules
At the same temperature, the average KE of all particles is the same
In a matter, not all the particles have the same KE at a particular temp, some above average energy, some below average
When temp ^, the average KE of particles ^, particles move at higher average speed average (KE of particles ∝ temp)
Evaporation
-change of state, liquid > vapour, at any temp w/o reaching bp
evaporation requires (heat) energy (from surroundings), liquid molecules: random movement, faster molecules, large enough KE > overcome intermolecular attractive forces > leaves at surface > av KE of molecules remaining decreases > temp decreases, cooling effect
Factors affecting evaporation
Surface area of liquid; larger area, more evaporation
Temperature; higher temp, more evaporation, more KE to escape
Atmospheric pressure; atmospheric pressure , evaporation , escaped molecules less likely to be bound back into liquid
Draft over surface; stronger draft, move evaporation, molecules removed asa escape liquid
Humidity of surrounding air; more humid less evaporation
Nature of liquid; lower bp more evaporation
Evaporation;
- at all temp
-slow process
-at surface
-no bubbles formed
-temp may change
-heat from surroundings
Boiling;
-at fixed temp
-quick process
-in liquid
-bubbles formed
-temp constant
-heat from source
Expansion
Expansion of solids: temp ^, molecules gain energy > vibrate more vigorously > pushing each other further apart > increased movement = expansion, when object cooled > vibrations smaller > contracts (at absolute 0, molecules stop vibrating)
amt of expansion or contraction depends upon: 1temp change, 2size of object; larger > more expansion, 3type of material; diff metals expand differently for same temp rise (Al expands 3 times more than steel)
Uses of thermal expansion and contraction
-separating and joining materials; shrink fitting
axle too large for hole in wheel > axle contracts in liquid N2 > cold axle fits into wheel > axle: normal temp > tight fit
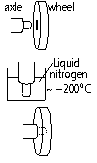
-Bimetallic strip; 2 strips of diff metals firmly fixed together: heated > 1 metals expands more > strip bends, cooled > bends other way (iron + brass, invar + brass, iron + copper)
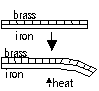
Uses of bimetallic strips
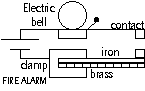
-fire alarm; fire > heat cause BM strip to touch contact > bell rings
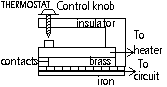
-thermostat; keep temp ~constant: too hot:breaks circuit > cools: closed circuit, control knob screwed down > higher temp breaks circuit
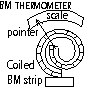
-bimetallic thermometer; 1 end fixed and other attached to pointer, when temp rises > BM spiral curls anti- clockwise > pointer moves
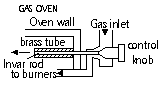
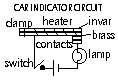
Examples
Bridges; rollers at one end allow movement, if not bridge buckle under hot weather
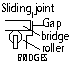
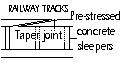
Railway tracks; No allowance; very hot day > bent and severely damaged, taper joint allows expansion and smoother ride, Concrete sleepers used for central portion > withstand large temp changes
Overhead power lines and telephone wires; Summer > expand, sag, winter > contract, tighten
Thick glass containers; Glass break due to localised expansion of glass, inside expands while outside doesnt as glass is a bad heat conductor, to avoid this, pyrex is used > expands v.little
Bottles; air space above liquid in bottle, allows expansion of liquid, compressing air
Tyres; cold > too small for wheel, heated > fits wheel, contracts > tight fit


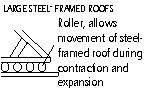
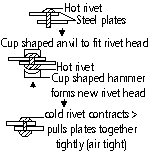
Expansion of Liquids
Liquids heated > expand (except water) more than solids
Expansion of water
-4 °C - 100°C > like normal liquids, heat > expand, cool > contract
-0°C - 4°C; cool > expands, heat > contract
(vol of fixed amt of H2O smallest at 4°C, max density at 4°C)

H2O at top of pond cools > contracts > denser > sinks, warmer less dense H2O rises > cooled at surface, when all H2O cools to 4°C circulation stops, temp of H2O at top < 4°C > less dense > stays of top > forms ice layer
when H2O freezes at 0°C > considerable expansion (H2O:100cm3 >ice: 109cm3) > ice at 0°C is less dense than H2O at 0°C (ice floats on H2O), in very cold weather, water pipes might burst
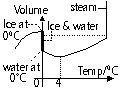
Expansion in gases
Very much larger expansion than liquid and gases
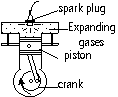
Petrol & air mixed > ignited > explode > the expanding gases push piston downwards into cylinder > drives the wheel of vehicle by rotating crank
Brownian motion
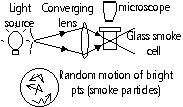
Observation
-smoke particles scatter light shining on them > appear as bright pts, smoke particles move continuously and randomly, larger particles > less agitation
Explanation
-fast moving air molecules (too small to see) bombard smoke particles unevenly on diff sides > irregular movement = Brownian motion (pollen grains in H2O > grains hit by H2O molecules)
Brownian motion provides evidence that molecules in a liquid or gas are always in continuous, random motion for the kinetic molecular model of matter
Diffusion- spreading of fluids from high > low conc due to molecular motion
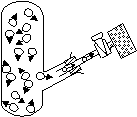
Observation
-within a few minutes brown bromine > diffusion tube
-vacuum: almost instantaneous movement of bromine > diffusion tube
Explanation
-random movement of air and bromine, bromine: inlet tube (high conc) > diffusion tube (low conc), air molecules: diffusion tube > inlet tube
-vacuum: no molecules to obstruct path > shows movement of molecules very fast (~200 m/s)
Brownian motion and diffusion gives evidence that particles are in continuous random motion
Pressure in gases
due to molecules repeatedly hitting container walls many times a second > exerts a force & pressure on walls (pressure = force/ area), temp ^ > av.speed of molecules ^> pressure ^, vol decrease > dist bet collisions decrease & hits per second ^ > pressure ^
Pressure-volume relationship of a gas(Boyles Law, temp constant)
Gas trapped in vertical tube > piston pushed inward > increase pressure
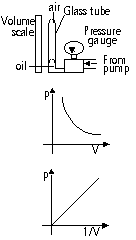
p ∝ 1/V
pV = constant
p1V1 = p2V2
p ∝ 1/V, as vol > # of molecules per unit vol x2 > molecules travel shorter dist to collide w/ wall > > # of collision x2 > pressure x2
Volume-temperature relationship of gas (Charles Law, pressure constant)
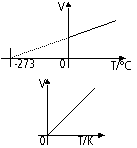
V ∝ T (absolute temp)
V/T = constant
V1 = V2
T1 T2
Absolute temp; unit-kelvin (K)
0K = -273°C, 273K = 0°C
0 K = absolute zero -lowest temp possible temp, molecules have lowest possible KE
Pressure-temperature relationship of gas (pressure law, volume constant)
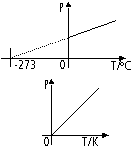
p ∝ T (absolute temp)
p/T = constant
P1 = P2
T1 T2
Ideal gas equation
pV = constant
T
p1V1= p2V2
T1 T2
Thermal physics: part 2
Waves & Sound
Radioactivity
Measurements In Physics
Forces
Energy
Pressure
Optics
Magnetism
Electrostatic Charging
Electricity
Back to 'O' level notes index
Back to notes index