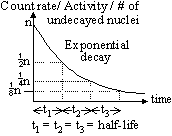
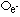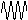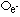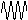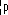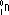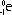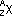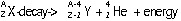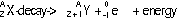| Properties | α-particles | β-particles | γ-radiation
|
|---|
| nature | positive particle: helium nucleus, a stream of He nuclei (~7000 times mass of an electron) | stream of high energy e-s, formed from nucleus decay | electromagnetic waves, v.short wavelength
|
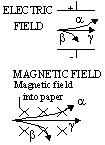
| affected by electric and magnetic fields | Yes | yes, bent strongly | no
|
| penetration | stopped by paper or skin or 6cm of air | stopped by 3mm of Al | reduced but no stopped by lead (50%)
|
| causes ionisation | Strongly | weakly | very weakly
|
| dangerous | yes | yes | yes
|
| speed | 10% speed of light | 50% speed of light | speed of light
|
| detection | Photographic film, cloud chamber, G-M tube, spark counter, gold-leaf electroscope | photographic film, cloud chamber, G-M tube | photographic film, cloud chamber, G-M tube
|