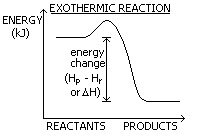Chem7
RATE AND HEAT OF REACTIONS + REVERSIBLE REACTIONS
RATE OF REACTION
Fast-explosions, slow-baking cakes, formation of oil + coal
Depends on particles colliding and having enough energy (break bonds/ forces of attraction)
Concentration (solutions)
-high conc. faster > more collisions
Pressure(gas) [low pressure > lower b.p.]
-high pressure faster > less space > more collisions
Particle Size(solids)
-larger SA, faster > more area to collide
Temperature(increase in 100°C, 2x reaction rate)
-high temp, faster, more speed > more collisions, more energy > break bonds/ forces of attraction
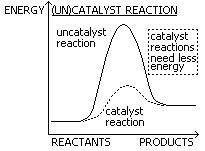
Catalysts
-chemical that speed up (or slow down) reactions, not used up in reaction, remain unchanged, reaction requires less energy
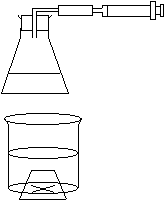
Measuring Speeds of Reaction
-measure vol. of gas produced or escaping
-measure mass of reactant and products
-measure time taken for cross to disappear (ppt.)
HEAT OF REACTION
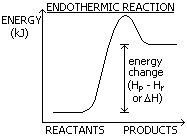
There is a given amount of energy (enthalpy) in reactants (Hr) and products (Hp)
Change in energy (kJ) = Hp - Hr or ΔH
Energy taken in- endothermic or given out- exothermic
EXOTHERMIC
Enthalpy- lost/ given out > temp. increase
Reactants more enthalpy
ΔH:-tive
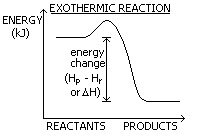
ENDOTHERMIC
Enthalpy- gained/ taken in > temp. decrease
Products more enthalpy
ΔH:+tive
Bond breaking-uses energy, products more energy > endothermic
Bond forming-loses energy, reactants more energy > exothermic
Photosynthesis- endothermic- light used to make food (carbohydrates), 6CO2 + 6H2O + light > C6H12O6 + 6O2
Photography- endothermic- light abosrbed by Ag+ ions to make silver (dark areas on film) 2AgCl + light > 2Ag + Cl2
Fuels- exothermic- burning of fuels, wood, coal, oil, gas, petrol, etc, from wood and plant materials, 2C8H18(petrol)+ 25O2 > 18H2O + 16CO2 + E
Hydrogen- exothermic- rocket fuels, liquid H2 & O2 mixed burnt: 2H2 + O2 > 2H2O + E
Nucluer Energy- exothermic- produces large amount of energy, heat H2O > steam > elec. Energy, waste products > pollution
Batteries (Electric cells)- exothermic- 2 dif. metals in an electrolyte generate elec. chem. energy > elec. energy, more reactive metal > ions > e- move to less reactive metal > elec. (further metals apart in reactivity series > larger voltage)
REVERSIBLE REACTIONS
NaCl (s) → Na+ (aq) + Cl-(aq)
Na+ (aq) + Cl-(aq) → NaCl (s)
NaCl (s) ↔ Na+ (aq) + Cl-(aq)
→ forward reaction, ← reverse reaction, ↔ reversible reaction
reversible reaction = a reaction where there is a forward and reverse reaction
equilibrium-constancy of macroscopic properties in a closed system: when speed of forward reaction = speed of reverse reaction > conc of products and reactants, constant, requires a closed system so chemicals can't enter or escape which will lead to change in equilibrium
CONCENTRATION: reactants conc increased > forward reaction favoured
PRESSURE(gases): side w/ more # of moles more affected
TEMPERATURE: side w/ energy more affected
PARTICLE SIZE: no effect on equilibrium, only speeds up to equilibrium
CATALYSTS: no effect on equilibrium, only speeds up to equilibrium
The Periodic Table
Metals
Organic Chemistry
Non-metals
The Particulature Nature of Matter
Experimental Techniques
Atoms, Elements and Compounds
Stoichiometry + Mole Concept
Acids, Bases and Salts
Chemical Reactions + Electrolysis
Back to 'O' level notes index
Back to notes index