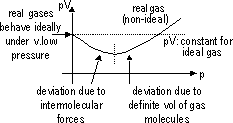
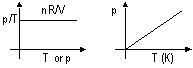| PARTICLES IN SOLID
| PARTICLES IN LIQUID
| PARTICLES IN GAS
|
| closely packed together + attracted by strong forces of attraction
| quite close together, not as close together as in solid, attracted by forces weaker than in solid
| far apart, attracted by weaker forces of attraction
|
regularly arranged in lattice structure

| not orderly arranged
| not orderly arranged, move about randomly

|
| least KE > not enough to overcome attractive forces > particles only rotate & vibrate at fixed positions
| enough KE to overcome attractive forces while in close proximity > particles rotate, vibrate & translate from 1 place to another
| large KE > overcome attractive forces > particles vibrate, rotate & translate from 1 place to another freely
|