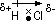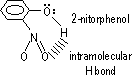CHEMICAL BONDING part 2
Bond length: dist bet 2 nuclei of atoms in a covalent bond (nm)

Bond strength: bond energy / bond enthalpy/ bond dissociation energy - min energy to dissociate 1 mole of covalent bonds in gaseous molecules to form gaseous atoms
-molecule w/ > 1 covalent bonds of same type > bond energy = average value (bond broken 1 by 1)
shorter bond length > greater bond strength
Breaking covalent bonds: homolytic fission/ heterolytic fission
Homolytic fission:- cov bond broken > free radicals (atoms w/ upaired e-s, retain valence e-s used in bond)
Heterolytic fission:- cov bond broken > ions (more electronegative atom will take valence e-s > anion)
bond enery: for homolytic fission of covalent bonds only
Dipole- separation of charge formed in a cov bond when displacement of bond pair of e-s towards more electronegative atom occurs, denoted by
' '
'
polar bonds have dipoles, non-polar bonds: no dipoles
for a covalent bond to be polar: bonds must be polar & molecule structure must not be symmetrical (symmetrical: dipoles cancel each other > no resultant dipole)
strength of dipole measure as dipole moment (units: Debye)
Non-polar molecules:
covalent bonds are non-polar if
-bonds are non-polar /
-bonds are polar, but molecule has center of symmetry so resultant dipole moment = 0
Fajan's rule & polar covalent bonds:
bond bet metal + non-metal atom usually ionic. Some metal chlorides- polar covalent due to polarization of anion by cation
Fajan's Rule: cation w/ small ionic radius & high charge density + large anion > bonds predominantly covalent (polarization of anion by cation = cation attracts valence e-s from anion > orbital overlapping > e-s shared > polar covalent bond)
Physical properties of covalent compounds:-
-compounds of simple molecular structure: low mp & bp (weak intermolecular forces of attraction broken)
-compounds of giant covalent structure: high mp & bp (many strong cov bonds need to be broken)
-consist of molecules / atoms, no ions / e-s > non-electrolytes (some dissolve in water > electrolytes, HCl)
-most insoluble in polar solvents (water) [propanone(acetone) + ethanol soluble in water]
-most w/ simple molecular structure: soluble in non-polar organic solvents (I in CCl4, S(s) in CS2/ methylbenzene)
Metallic Bonding (in metals)
metals: lattice structure w/ +ve metal ions (metal cores) in a sea of mobile valence e-s (can move throughout structure under electric field and attracts + holds metal ions together in solid)
metallic bond = electrostatic force of attraction bet mobile valence e-s & +ve metals cores
Physical properties:-
-high mp, bp & density (+ve metals ions held strongly together by metallic bonds)
-conduct elec (good conductors of elec) (valence e-s mobile > free to move under electric field)
-usually shiny & lustrous (mobile valence e-s absorb light energy absorbed > give out light w/ same wavelength > reflection of light)
-malleable & ductile (+ve metal cores attracted from all directions by -ve valence e-s > able to move about)
Intermolecular Forces of Attraction
-intermolecular forces of attraction are all weaker than bonds
-3 types: H bonding, permanent dipole-dipole attraction, temp induced dipole-dipole attraction
Hydrogen bonding (bet polar molecules w/ hydrogen atom & electronegative atom)
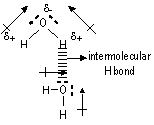
H bond: permanent dipole-dipole attraction bet H atom (attached to a very electronegative atom- usually N, F, O) & electronegative atom of another molecule
(H bond: special type of permanent dipole-dipole attraction)
Conditions for H bond:
-molecule must have H atom bonded to v. electronegative atom
-the electronegative atoms must have at least 1 lone pair
Types of H bond: intermolecular (bet two diff molecules) & intramolecular (within same molecule)
Intermolecular H bonding; occurs in: H2O, HF, NH3, coiled double helical structure in DNA, organic compounds w/ functional groups
Functional groups:
-OH (hydroxil group) (alcohol: R-OH)
-NH2 (amino group) (amine: R-NH2)
-COOH (carboxylic group)(organic acid: R-COOH)
Effects of intermolecular H bonding:
-increases mpt + bpt (H bonds strong > more energy needed)
-increases solubility in water (subs forms H bonds with H2O molecules) (NH3: v.soluble) (organic cpds don't usually dissolve in H2O, but methanol, ethanol & propanone-acetone do: formation of H bonds w/ H2O molecules)
(intermolecular H bonding causes dimerization of molecules: 2 molecules join together > larger molecule aka dimer, organic acids dissolved in solvent-benzene > dimers w/ RMM 2x original value)
Intramolecular H bonding;
(bet 2 adjacent groups in same molecule, groups usually -OH, -NH2, -COOH)
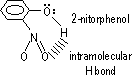
Compounds w/ intermolecular H bonding compared to compounds w/ intramolecular H bonding
Intermolecular H bonding:
-higher mp & bp (melting & boiling concerns breaking intermolecular bonds, intramolecular H bond cpds have only Van der Waals forces bet molecules)
-higher density (stronger intermolecular H bonds pack molecules more closely together than the weak Van der Waals forces in intramolecular H bond cpds)
-less soluble in water (strong intermolecular H bonds prevent dissociation of molecules, intramolecular H bonding: intermolecular H bonds w/ water prefer over Van der Waals forces bet molecules of compound)
Permanent dipole-dipole attraction (bet polar molecules)
-due to permanent dipoles in polar molecules> permanent attraction bet +ve & -ve side of diff molecules
Temporary induced dipole-dipole attraction aka Van der Waals force (bet non-polar molecules)
-when non-polar molecules are close together, bond pair(s) of e-s repel each other > temp displacement of e- pair(s) > temp dipole > temp attraction (very weak)
-also occurs due to continuous motion of e-s > instantaneous charge of a molecule which induces neighbouring molecules > molecules attract one another
Magnitude of Van der Waals force increases w/;
-RMM (more e-s > stronger dipoles > stronger attraction)
-decrease in dist bet molecules (less dist > more attraction)
(branched molecules that are not straight-chained have greater av.dist bet molecules > weaker VDW forces)
VDW force ∝ m1m2/d2
[m1 & m2 = RMM of molecules, d = dist bet molecules]
Magnitude of intermolecular forces of attraction:
H bond > permanent dipole-dipole attraction > temp induced dipole-dipole attraction
Chemical bonding part 1
Back to 'A' level notes index
Back to notes index

 '
'