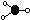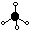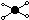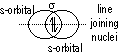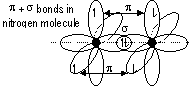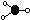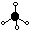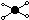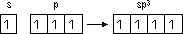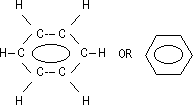CHEMICAL BONDING
Chemical bonds- electrostatic force of attraction bet atoms, molecules or ions
Chemical bonds- ionic, covalent (-dative/coordinate bonding), metallic, intermolecular force of attraction (-hydrogen bonding/permanent dipole-dipole bonding/temporary induced dipole-dipole attraction)
Ionic bonding
Ionic/electrovalent bond = electrostatic force of attraction (strong) bet +ve ions (cations) & -ve ions (anions)
cations/anions: from atoms of electro(positive/negative) elements by (losing/gaining) valence e-s > stable inert gas config (cations: usually metals, anions: usually non-metals)
Physical properties:-
-made of only ions attracted by strong ionic bonds, solid ionic compounds- giant ionic structure, usually crystalline
-usually solids at rtp, have high m.pt, b.pt & density (due to strong ionic bonds)
-as soild: don't conduct elec (ions not mobile), as molten liquid/ when dissolve in aqueous sol: conduct elec > liquid/sol = electrolytes (charged ions free to move)
-usually soluble in polar solvents(water = universal polar solvent) but not in non-polar organic solvents (benzene, ether,..)
Covalent bonding
Usually formed bet atoms of non-metals
Formed by pairing of valence e-s by atomic orbitals overlapping, e- pairs shared > inert gas config
(expansion of Octet rule: elements w/ 3d orbitals, >8 e-s in valence shell)
σ -bond: formed by overlapping of orbitals along linear lie joining nuclei of atoms (end-on overlapping)
(orbitals: s + s, s + p, p + p, s + hybridized, p + hybridized, hybridized + hybridized)
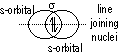
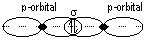

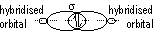
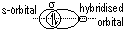
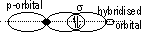
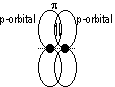
π -bond: formed by side-way overlapping of orbitals bet p orbitals [double- (σ + π)/triple- (π, π + σ) bonds] (weaker than σ -bond)
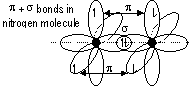
Shapes of covalent molecules
Electron pairs repulsion theory (valence shell electron pairs repulsion theory- VSEPR theory)
-used to predict shape of covalent molecules or ions w/ covalent bonds
rules: -magnitude of repulsions bet: (lone + lone) pair > (lone + bond) pair > (bond + bond) pair
-electron pairs in molecule are situated as far apart as possible > min e- pair repulsion
| molecule
| shape
| bond pairs
| lone pairs
| angle bet atoms
|
| linear
| 
| 2
| 0
| 180°
|
| trigonal planar
| 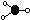
| 3
| 0
| 120°
|
| tetrahedral
| 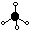
| 4
| 0
| 109.5° / 109°28'
|
| triangular pyramidal
| 
| 3
| 1
| 107°
|
| bent
| 
| 2
| 2
| 104.5°
|
| trigonal bipyramidal
| 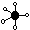
| 5
| 0
| 90°, 120°
|
| t-shape
| 
| 3
| 2
| 90°
|
| octahedral
| 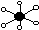
| 6
| 0
| 90°
|
| square planar
| 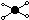
| 4
| 2
| 90°
|
t-shaped molecule: interhalogen compounds
Electronegativity:- ability of atom of element to pull shared e- pairs in cov bonds towards itself
-greater electronegativity of central atom > greater bond angle
Dative/coordinate bond: lone pair of e-s from an atom forms a covalent bond w/ an atom/ion (shared e-s comes from 1 atom)
Hybridisation of atomic orbitals: mixing of orbitals of diff energy level > set of identical hybrid orbitals w/ same energy (electronic config of element changes from ground state to excited state before hybridisation)
sp3 hybridisation: 1 s-orbital + 3 p-orbitals > 4 sp3 hybrid orbitals (> tetrahedral), methane (cage-like molecule)
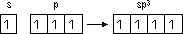
sp2 hybridisation: 1 s-orbital + 2 p-orbitals > 3 sp2 hybrid orbitals (> trigonal planar), ethane (planar molecule)
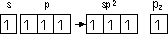
sp hybridisation: 1 s-orbital + 1 p-orbitals > 2 sp hybrid orbitals (> linear), ethyne (linear molecule: 1 σ-bond, 2 π-bonds)
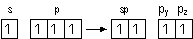
| Hybrid orbital
| Spatial distribution
| Shape
|
| sp
| linear
| 
|
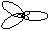
| sp2
| trigonal planar
| 
|
| sp3
| tetrahedral
| 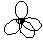
|
| sp2d
| square planar
|
| sp3d
| trigonal bipyramidal
|
| sp3d2
| octahedral
|
| sp3d3
| pentagonal pyramidal
|
Benzene: 6 C atoms > sp2 hybridisation > 6 trigonal planar orbitals overlap in a ring (σ bonds w/ H + C atoms), 6 2pz e-s (π e-s) can be delocalised over / under the 6 C atoms > sextet - ring of 6 delocalised π e-s
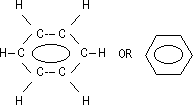
Chemical bonding part 2
Back to 'A' level notes index
Back to notes index