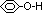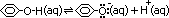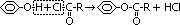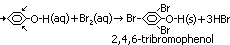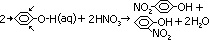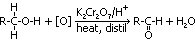AROMATIC HYDROXY COMPOUNDS
PHENOL
MF: C6H6O
functional group: phenolic group, C6H5OH
structural formula: 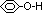
Physical properties
-at rtp, pure phenol = hygroscopic while solid w/ antiseptic smell (might be a little purple due to air oxidation)
(hygroscopic solid: solid absorbs water vapour in air > moist,
hydroscopic solid: absorbs so much water vapour > dissolve > colourless sol, eg FeCl3)
-corrosive
-partially soluble in small amt of water > cloudy emulsion (tiny immiscible oily pheol drops in water)
cloudy emulsion > colourless solution by: (1) dilution- add more H2O (due to more H-bonds w/ water), (2) heating- only temporary, on cooling > emulsion reforms
Chemical properties of phenols
-very weak acid (damped blue litmus: purple)
[cloudy aq. phenol + NaOH >(neutralization)> colourless sol: C6H5OH(aq) + NaOH(aq) > C6H5O-Na+(aq) + H2O(l)]
[C6H5O-: phenoxide ion]
[if dil H2SO4 / stream of CO2 added to sodium phenoxide sol, cloudy emulsion reforms:
C6H5O-Na+(aq) + H+(aq) > C6H5OH(aq) + Na+(aq)]
(phenol: much weaker than carbonic acid, H2CO3 & doesn't react w/ Na2CO3 to give CO2- distinguishes phenol from organic acids)
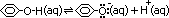
O atom's 2 π e-s interact w/ 6 π electrons of benzene ring > delocalisation of electrons stabilises phenoxide ion & weakens O-H bond (> phenoxide ion + H+) (ethanol: not acidic as ethoxide ion not stablised by delocalised e-s)
acidity of subs: based in pH / acid dissociation constant, ka (ka; not affected by changes in pH values) (greater ka: stronger acid) [pka = -lg ka (smaller pka: stronger acid)]
Reaction w/ Na
reagent: Na metal
condition: room temp
2C6H5OH(l) + 2 Na(s) > 2C6H5O-Na+ +H2
observation: -Na dissolves, -H2 gas
Alkanoylation
reagent: alkanoyl chloride, R-C(=O)Cl
condition: room temp
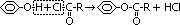
observation: -steamy white fumes of HCl, -sweet, fruity ester evolved
to form white solid phenylbenzoate: use alkaline sol of phenol
(C6H5OH + NaOH > C6H5O-Na+ + H2O
C6H5O-Na+ + C6H5-C(=O)Cl > C6H5-O-C(=O)-C6H5)
Reaction w/ aq Br
reagent: aq. Br
condition: room temp
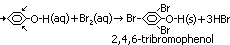
observation: immediate white ppt of 2,4,6-tribromophenol (excess phenol: red Br decolourised)
Nitration
reagent: conc HNO3
condition: room temp
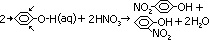
reagent: conc HNO3
condition: ~20°C
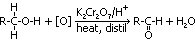
Test for phenol
-excess phenol + aq. Br > red Br decolourised + white ppt formed
-phenol + aq FeCl3 > violet colouration
(C6H5OH + PCl5 > C6H5Cl+ POCl3 + HCl: v.low yield ~ no reaction)
phenol is not oxidised
Aromatic hydroxy compounds part 1
Back to 'A' level notes index
Back to notes index