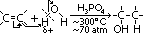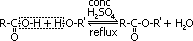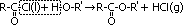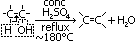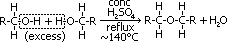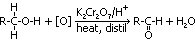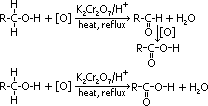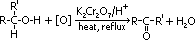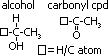ORGANIC HYDROXY COMPOUNDS
hydroxy cpds: contain -OH group
hydroxy cpds:
-alcohol (-OH attached to aliphatic C chain) (monohydric alcohol: 1 -OH, diol: 2 -OH, triol: 3 -OH)
-phenol (-OH attached to C atom of benzene ring)
Monohydric alcohols
general formula: CnH2n+2O or CnH2n+1OH [n=1,2,..]
functional group: hydroxy group, -OH
Types of alcohols
primary (1°) alcohol: C atom attached to -OH group has 2 H atoms attached
secondary (2°) alcohol: C atom attached to -OH group has 1 H atom attached
tertiary (3°) alcohol: C atom attached to -OH group has no H atoms attached
Isomerism in alcohols
CnH2n+2O:- general formula for alcohols(-C-OH) & ethers (-C-O-C-)
Industrial preparation
Fermentation of glucose to form ethanol
-Hydrolysis of starch: starch >(enzyme)> glucose
(C6H10O5)n + nH2O >(enzyme)> nC6H12O6
-Fermentation of glucose: glucose >(yeast)> ethanol + CO2
C6H12O6>(yeast)> 2CH3CHOH + 2CO2
ethanol produced is purified by distillation (bp: 78.5°C)
Catalytic Hydration of alkenes
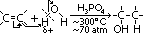
alkene + steam >(H3PO4, 300°C, 70 atm)> alcohol
Lab preparation
Hydrolysis of halogenoalkanes
R-X + NaOH(aq) >(heat, reflux)> R-OH + NaX
halogenoalkane + aqueous sodium hydroxide, heat under reflux > alcohol + sodium halide
Reaction between 1° amine & nitrous acid
reagent: sodium nitrite, NaNO2, acidified w/ dil HCl [NaNO2 + HCl > HNO2]
conditions: ice-cold temp (0°C-10°C)
R-NH2 + HNO2 >( ice-cold temp)> R-OH + H2O + N2 [amino group replace by hydroxy group]
observation: bubbles of gas appear (N2)
Physical properties
-characteristic smell
-lower members: colourless liquids, higher members: white waxy solids [as RMM ^ > VDW forces ^ > solid-like]
-compared to cpds w/ similar RMM, alcohols have higher mp + bp (H-bonds bet alcohol molecules)
-1st 3 alcohols completely soluble in water (H-bond bet water & alcohol molecule) (higher members: H-bonds bet alcohol molecules to strong to be overcome by H-bonding in water)
Chemical properties
Reactions:
-reactions involving -OH group (-reaction w/ Na, -conversion to halogenoalkane, -esterification)
-dehydration
-oxidation
-triiodomethane test
-combustion
Reaction involving -OH group
Reaction w/ electrophile metals (eg. sodium)
reagent: Na metal
condition: room temp
2R-OH(l) + 2Na(s) > 2R-O-Na+(aq) + H2(g)
observation: if excess alcohol -sodium metal dissolves > colourless liquid of sodium alkoxide (R-ONa), -effervescence of hydrogen gas
Conversion to halagenoalkane
-OH group replaced by halogen, used to prepare halogenoalkanes
Replacing w/ chloride group, Cl -
reagent: phosphorus (V) chloride, PCl5 (phosphorus pentachloride)
condition: room temp
R-OH + PCl5 > R-Cl + POCl3(l) + HCl(g)
observation: white fumes of HCl gas evolved > indicates presence of -OH group
Replacing w/ iodide group, I - or bromide group, Br-
reagent: red phosphorus + halogen, X (X = bromine / iodine)
condition: heat under reflux
2P(s) + 3X2 >(heat)> 2PX3 (phosphorus tribromide/ triiodide)
3R-OH + PX3 >(red P, X, heat, reflux)> 3R-X + H3PO3 (phosphorus (III) acid)
Formation of ester
Esterification
reagent: alkanoic acid + conc H2SO4
condition: heat under reflux
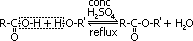
reaction is reversible, so conc H2SO4 added to favour forward reaction by absorbing water, conc H2SO4 is also a catalyst by providing H+(aq)
observation: if poured into beaker of H2O- ester floats on H2O surface > oily patches of sweet, fruity smelling ester
Alkanoylation/ Acylation of alcohol
reagent: alkanoyl chloride = acyl chloride, R(=O)Cl
condition: room temp
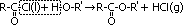
observation: -sweet, fruity smell of ester, -steamy white fumes of HCl gas
Dehydration of alcohol to form alkene
reagent: conc H2SO4
conditions: -conc H2SO4 in excess, -heat under reflux, temp: ~180°C
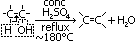
Dehydration of alcohol to form ether
reagent: conc H2SO4
conditions: -alcohol in excess, -heat under reflux, temp: ~140°C
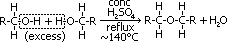
Oxidation of alcohols
reagent: oxidising agent- acidified potassium dichromate(VI), K2Cr2O7/H+ [or KMnO4/H+]
observation: excess alcohol- orange K2Cr2O7 > green (Cr3+(aq) formed) [purple KMnO4 decolourised (v.pale pink)]
1° alcohol:
conditions: (1)heat & distil > aldehyde obtained, (2)heat, reflux > aldehyde >(further oxidised) >alkanoic acid
formation of aldehyde:
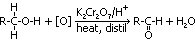
formation of alkanoic acid:
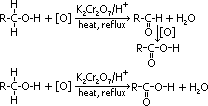
2° alcohol:
condition: heat under reflux
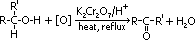
3° alcohol: resistant to oxidation
Triiodomethane test
only works for alcohols / carbonyl cpds w/ following structure
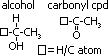
reagents: -aq. I2 sol [I2 dissolved in KI(aq)], -NaOH(aq)
condition: warm mixture
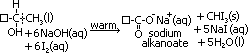
observation: yellow ppt w/ antiseptic smell of CHI3
(R-CH(OH)CH3: -CH3 part forms triiodomethane, R-CH(OH)- part oxidised > sodium salt of alkanoic acid)
(ethanol: only 1° alcohol that tests +ve)
[triiodomethane test:
2NaOH + I2 > NaI + NaOI + H2O
R-CH(OH)CH3 + NaOI > R-C(=O)CH3 + NaI + H2O
R-C(=O)CH3 + 3NaOI > CHI3 + R-C(=O)O-Na+ + H2O]
Combustion
alcohol(ethanol) burns in air w/ pale blue flame (clean + non-luminous) due to oxygen content (-OH group)
Aromatic hydroxy compounds part 2
Back to 'A' level notes index
Back to notes index