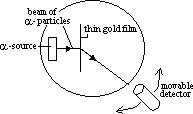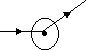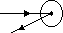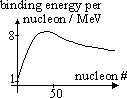ATOMIC STRUCTURE
Rutherford's α-particle scattering experiment
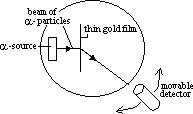
-evidence of nucleus in atoms
-gold film bombarded w/ α-particles > most particles pass straight through, some deflected through small angles, some deflected large angles (even backwards)
explanation
-led to atom model: heavy +ve charged nucleus at center, w/ v.light –ve charged electrons in orbit around it
explanation
(1)
pass straight through- atom almost 'empty' w/ v.small nucleus
(2)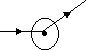
deflected slightly- due to repulsive F of +ve nucleus & +ve α particles
(3)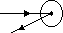
deflected backwards- hit nucleus, bounce back
Atomic measurement
unified atomic unit (µ) used to measure mass of atomic particles [µ-very close to mass of proton / neutron]
proton = 1.00728µ, neutron = 1.00867µ, electron = 0.00055µ
for convenience; 1µ = mass of carbon-12 atom/12 = 1.66 × 10-27kg
Elements, nuclides, isotopes
most elements: a sample is mixture of diff versions
nuclide: any particular version of atom, AZX
nucleon #, A: total # of nucleons (ie, protons & neutrons)
atomic/proton #, Z: total # of protons (= total # of electrons for neutral atom)
isotopes: nuclides w/ same proton # but diff nucleon # (same electron arrangement > same chem. properties)
[lithium, Li: element, Li-6: nuclide, Li-7: nuclide, Li-6 & Li-7: nuclide]
Nuclear energy
from Einstein's theory of relativity => E has mass
=> obj gain / lose E > gain / lose mass
ΔE = Δmc2 [rest mass: mass of obj at rest]
c: v.large => Δm v.small [nuclear reactions prod significant Δm]
Mass defect
-difference in mass of atom & sum of mass of particles in atom
-due to particles being bound together in atom by strong nuclear force
-to separate particles work is done => particles have less PE than if free => smaller mass
-all nuclides have mass defect (except hydrogen-1, due to only 1 proton)
Binding energy- energy needed to split nucleus into separate nucleons (energy equivalent of mass defect)
nuclear binding energy = mass defect × c2
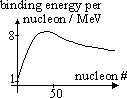
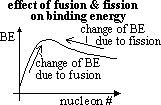
stability of nucleus proportional to binding energy, BE per nucleon
energy released when nucleons rearrange to obtain higher BE
[radioactive decay: unstable nuclei decay to form stable products => energy released
alpha decay: most energy in form of KE of alpha-particles]
Nuclear reactions
-fission & fusion both release energy
-fission: nucleus split => form nucleas w/ higher BE (near peak of graph)
-fusion: nuclei join to form heavier nucleus
transmutation
-1 element changes into another
-can occur when atom bombarded by particles
Mass excess
-when nucleus bombarded by energetic particle => particle absorbed & nucleus exists in highly excited state > nucleus disintegrates & ejects a particle > becomes diff nucleus
-mass excess = increase in total rest mass of particles after nuclear reaction
-due to large KE of bombarding particles, decrease in KE = increase in mass
Nuclear fission
-heavy nucleus splits into (i)2 nuclei of about the same mass & (ii)neutron(s)
-rarely occurs spontaneously; usually when neutrons hit nucleus
-E released: mostly as KE of heavy particles
-E released is v.large, ~50 million times per atom greater than E released from chem. reactions (burning)
Nuclear fusion
-harder to achieve compared to fission due to repulsive F bet +ve nuclei (need to be heated to > 108K)
-Sun gets energy from fusion of hydrogen
Back to 'A' level notes index
Back to notes index