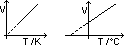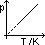IDEAL GAS
GAS LAWS(T- temp/K, p- pressure/Pa, V- vol/m3)
Boyle's law
T const => p ∝ 1/V (const mass)
pV = k

Charles's law
p const => V ∝ T (const mass)
V/T = k
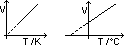
Pressure law
V const => p ∝ T (const mass)
p/T = k
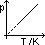
general gas law/ eqn of state: pV/T= k
-accurate for low pressure (high pressure: gas behaviour deviates)
-concept of ideal gas derived => ideal gas obey eqn under all conditions
Ideal gas eqn; pV/T = k => p1V1/T1 = p2V2/T2
pV/T ∝ n (n: # of moles) => pV/T = nR (R = molar gas const)
pV = nRT
n = (N/NA)RT
R/NA = k = Botlzman's constant (gas const for individual molecule)
pV = NkT
Assumptions of kinetic theory of gases
-any gas consists of a v.large # of molecules
-molecules of gas in rapid, random motion
-collision of gas molecules w/ each other and walls of container are perfectly elastic
-intermolecular forces of attraction are negligible
-duratn of collision negligible compared to time interval bet collisions
-vol of gas molecules negligible compared to vol of container
Kinetic calculation of pressure
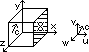
cube of side l containing N molecules
-each molecule: -mass, m, -velocity at any intstant, c
c resolved into u, v, w (components in x, y, z dirn)
c2 = u2 + v2 + w2
considering x-component, u
F exerted by face X during collision
momentum change on impact = mu - (-mu) = 2mu
time bet collisions w/ face X = 2l/u
freq of collisions w/ face X, n' = 1/(2l/u) = u/2l
at X: momentum change per second = F (Newton's 2nd law)
F = n' × momentum change per collision = u/2l × 2mu = mu2/l
pressure on X = F/A = (mu2/l)/l2 = mu2/l3 = mu2/V
each molecule diff vel => diff mag of compenent in x dirn, u1, u2,.., un
p = m(u12 + u22 + .. + un2)/V
<u2> = (u12 + u22 + .. + un2)/N
p = Nm<u2>/V
since large # of molecules => <u2> = <v2> = <w2>
<c2> = <u2> + <v2> + <w2>
<u2> = <c2>/3
<c2> = mean square speed
p = Nm<c2>/3V
pV = Nm<c2>/3
pV: macroscopic
Nm<c2>/3 : microscopic
Nm/V = M/V = denisity, ρ
p = ρ<c2>/3
rms speed, crms = √<c2> = √ [(c12 + c22 + .. + cn2)/N]
p = ρ<c2>/3 => crms represents av speed of all molecules to define a particular state (giving rise to a particular pressure at a particular temp)
p = (Mm/Vm)<c2>/3 Mm: molar mass, Vm: vol of 1 mole of gas
pVm = Mm<c2>/3 = RT (n = 1) => <c2> = 3RT/Mm
crms = √(3RT/Mm)
pV = NkT
pV = Nm<c2>/3
kT = m<c2>/3 => KE = m<c2>/2 = 3kT/2
av KE of translation of molecules ∝ T (T: abs temp, K)
Back to 'A' level notes index
Back to notes index