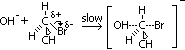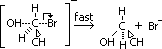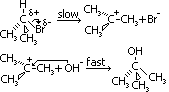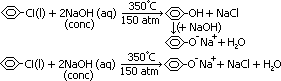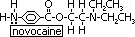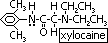HALOGENOALKANES
general formula: CnH2n+1X [n=1,2,.. X = F,Cl,Br,I]
functional group: halogen group; F: fluoro-, Cl: chloro-, Br: bromo-, I: iodo-
halogenoalkanes: primary(1°), secondary(2°), tertiary(3°)
primary: C atom has 2/3 H atoms attached to it
secondary: C atom - 1 H atoms attached
tertiary: C atom has no H atoms on it
Physical properties
-lower members w/ low RMM > gases at rtp(small VDW forces) [CH3Cl, CH3Br, CH3CH2Cl]
higher members > liquid / solid [CH3I]
-characteristic smell
-insoluble in polar solvents(water), soluble in non-polar solvents
Chemical properties
-Nucleophilic substitution: hydrolysis, conversion to alkanonitrile, conversion to amine
-Elimination: dehydrohalogenation to form alkene
Nucleophilic substitution
C-X bond: polar, X more electronegative than C atom > X: nucleophilic, C: electrophilic

Alkaline hydrolysis to form alcohol
reagent: NaOH(aq)
condition: heat under reflux
equation: -C-X + NaOH(aq) >(reflux)> -C-OH + NaX
if products neutralise w/ acid (NaOH reacts w/ Ag+), then add AgNO3: silver halide ppt, AgX, X= Cl-white ppt, Br-pale yellow ppt, I-yellow ppt
Mechanism of nucleophilic substitution in alkaline hydrolysis of bromoethane
-nulceuphile attacks +ve C atom of C-Br bond (due to polarisation) > transition state complex formed
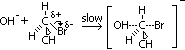
-carbanion decomposed to give ethanol
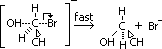
Energy diagram of alkaline hydrolysis of bromoethane
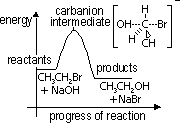
slow step mechanism involves 2 subs: OH- & CH3CH2Br > a 2nd -order reaction, rate eqn: rate = k[CH3CH2Br][OH-], k = rate const. [ ] = molar conc of reactant
(hydrolysis of 3° halogenoalkane undergoes a diff nuclephilic subs mechanism: carbocation intermediate)
Mechanism of 3° halogenoalkane
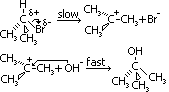
Energy diagram of reaction

1st-oder reaction involving only (CH3)3Br, rate eqn: rate = k[(CH3)3Br]
Conversion to alkanonitile
reagents: aqueous ethanolic potassium cyanide, KCN (KCN dissolved in mixed solvent of ethanol + water, ethanol: solvent for halogenoalkane, water: solvent for KCN)
conditions: heat under reflux
equation: -C-X + KCN (ethanolic) >(reflux)> -C-CN + KX
[X replaced by cyano group, -CN > C atom increased by 1]
alkanonitrile can be converted to amine (by reduction) or acid (by hydrolysis) then to alcohol
RCN + 4[H] >(reduction: LiAlH4/ dry ether, reflux)> RCH2NH2 (amine)> alcohol
RCN + 2H2O >(hydrolysis: dil HCl, heat)> RC(=O)OH (acid) + NH4Cl > alcohol
Conversion to amine
reagent: ethanolic ammonia (ammonia dissolved in ethanol)
condition: heat in sealed vessel (to prevent escape of gas)
equation: RX + NH3 (ethanolic) >(heat in seal vessel)> RNH2 + HX [HX(acidic) + NH3 > NH4X]
primary(1°) amine: 2 H atoms attached to N atom
secondary(2°) amine: 1 H atoms attached to N atom
tertiary(3°) amine: no H atom attached to N atom
(excess halogenoalkane: mixture of 1°, 2°, 3°, quarternary alkylammonium salt)
CH3CH2Br (excess) + 2NH3 > CH3CH2-NH2 + NH4Br
CH3CH2Br + CH3CH2-NH2 (nulceophilic) > NH(CH2CH3)2 + HBr
CH3CH2Br + NH(CH2CH3)2 (nulceophilic) > N(CH2CH3)3 + HBr
CH3CH2Br + N(CH2CH3)3 (nulceophilic) > N(CH2CH3)4+Br-
Elimination
Dehydrohalogenation to form alkene
hydrogen halide molecule, HX removed from halogenoalkane to form alkene
reagent: ethanolic NaOH
condition: heat under reflux
equation:

HALOGENOARENES (aryl halides)
cpds w/ halogen atom attached to C in benzene ring
molecular formula: C6H5X [X = halogen]
formation of halogenoarenes
C6H6 + X2 >(Fe)>C6H5X + HX
Chemical properties
halogenoarenes: unreactive compared to halogenoalkanes
due to CX bond in halogenobenzene less polar than CX bond in halogenoalkane
p π-electrons of halogen interact w/ 6 π-electrons of benzene ring > delocalised > CX bond stronger + less polar
halogenoalkane doesn't react w/ dil NaOH(aq) even under reflux > need drastic conditions (conc NaOH(aq), heat under pressure + temp-350°C, 150atm)
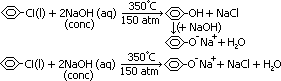
phenol, C6H5OH: weak acid, not alcohol, reacts w/ NaOH > ionic sodium phenoxide salt, C6H5O-Na+
CFC = ChloroFluoroCarbons (chlorofluoroalkanes)
C-F bond strongest halogen-C bond > stable (C-Cl bond 2nd strongest) > chlorofluoroalkanes > stable & unreactive org cpd
Properties of CFC
-odourless
-low toxicity
-chemically unreactive
-non-flammable
-low bp
-good organic solvent
Uses of CFC
Refrigerant: dichlorodifluoromethane (CCl2F2) (bp= -30°C) in air-conditioners or refrigerators. has high RMM w/ large intermolecular VDW forces > liquefied easily by compression & cooling (Joule-Thompson's effect), it is also non-corrosive & non-toxic
Aerosal propellant: dichlorodifluoromethane (CCl2F2) produces spray of 'mist' containing active subs eg perfume, insecticide, paint,..
Solvent: 1,2-dichloro-1,1,2,2-tetrafluoroethane (CF2ClCF2Cl) (bp =48°C, volatile (l) at rtp)- degreasing solvent for machinery parts & electronic circuit components
Ozone depletion
formation of ozone(O3):
O2(g) >(UV from sun)> 2O(g) (oxygen atoms)
O(g) + O2(g) > O3(g)
photochemical reaction bet CFC and ozone:
CF2Cl2 > CF2C• + Cl• (chlorine atom-FR)
Cl• + O3 > •OCl + O2(g)
•OCl + O > Cl• + O2
(Cl• reproduced > continue reaction w/ other ozone molecules)
Substitutes for CFC
-CO2,N2, propene gas, methylpropane as aerosal propellants
disadvantages: CO2, N2: don't produce fine 'mist', propane: flammable
-HCFC-hydrochlorofluorocarbon (CHF2Cl): halogenoalkanes w/ H atoms, H atoms make C-Cl bond more reactive > HCFC destroyed before reaches stratosphere to deplete ozone
Uses of other halogenated alkanes
-in fire extinguishers: BCF (halon 1211), bromochlorofluoromethane (CF2ClBr) in fire extinguishers for oil, gasoline, electric and chemical fires
-anaesthetic: fluothane or halothane before surgery, 2-bromo-2-chloro-1,1,1-trifluoroethane (CF3CHBrCl)
non-halogenated anaesthetic: N2O- laughing gas-dinitrogen oxide, novocaine, xylocaine
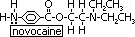
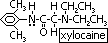
Back to 'A' level notes index
Back to notes index