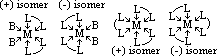
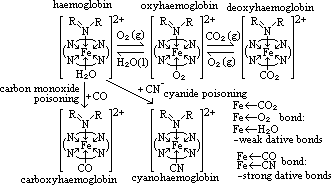
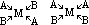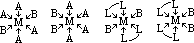| Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
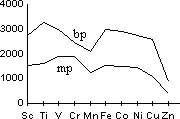
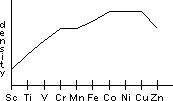
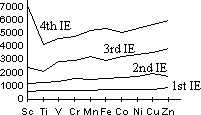
| Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
| +1 | +1 | +1 | +1 | +1 | +1 | +1 | +1 | ||
| +2 | +2 | +2 | +2 | +2 | +2 | +2 | +2 | +2 | |
| +3 | +3 | +3 | +3 | +3 | +3 | +3 | +3 | +3 | |
| +4 | +4 | +4 | +4 | +4 | +4 | +4 | |||
| +5 | +5 | +5 | +5 | +5 | |||||
| +6 | +6 | +6 | |||||||
| +7 | [in bold: important oxid state] | ||||||||
| water: aqua- | chloride: chloro- | oxide: oxo- | carbon monoxide, CO: carbonyl- |
| ammonia: ammine- | bromide: bromo- | cyanide, CN-: cyano- | sulphate, SO42-: sulphato |
| hydroxide: hydroxo- | iodide: iodo- | nitrite, NO2-: nitro- | C2O42-: oxalato- / ethanedioate- |
| edta4-: edta- | fluoride: fluoro- | ethane-1,2-diamine, en: ethane-1,2-diamine |
| metal | metal as cation | metal as anion | metal | metal as cation | metal as anion |
| copper | copper | cuprate | vanadium | vanadium | vanadate |
| iron | iron | ferrate | titanium | titanium | titanate |
| zinc | zinc | zincate | manganese | manganese | manganate |
| cobalt | cobalt | cobalate | nickel | nickel | nickelate |
| chromium | chromium | chromate |