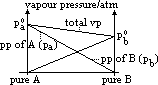
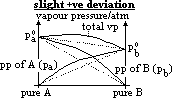
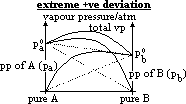
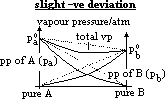
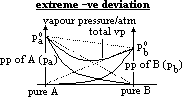
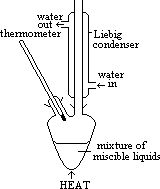
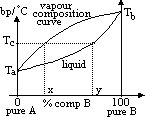
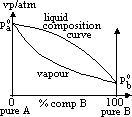
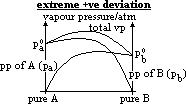
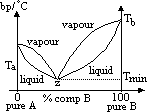
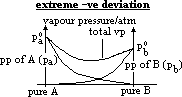
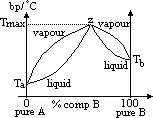

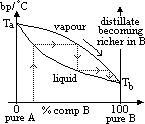

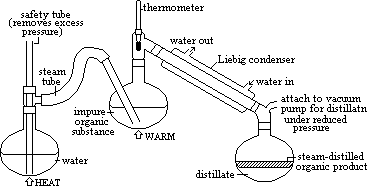
| Advantages | Disadvantages |
| decompostn less likely (distil at temp much lower than bp) | only small amt of liquid obtained c/o to large amt of water |
| only other sub used is water (cheap, safe, easily obtained) | org liq needs to be separated from water in distillate |
| process relatively simple, easily scaled up for industry | if org liquid partially miscible > emulsion formed > separatn difficult |