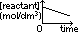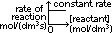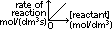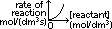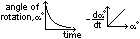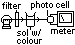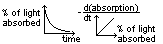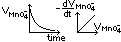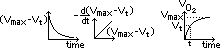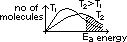REACTION KINETICS
rate of reaction: change of molar conc of reactant left or product formed per unit time [mol/(dm3time)]
rate eqn: rate = k[A]m[B]n, k = rate const, [] = molar conc of reactant left, m/m = order of reaction wrt A/B, m + n = overall order
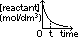 rate of reaction = -gradient of [reactant]-time graph
rate of reaction = -gradient of [reactant]-time graph
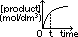 rate of reaction = -gradient of [product]-time graph
rate of reaction = -gradient of [product]-time graph
rate equation: expression that relates rate of reaction to (1)rate constant, (2)orders of reaction of reactants & (3)molar conc of reacts left at particular time
-rate equation determine experimentally (also based on stoichiometry of slow step / rate-determining step mechanism in a reaction, or from eqn if single-stepped reaction; slow step- A2 + 2B2 > 2AB2 > rate = k[A][B]2[C]0)
order of reaction: order of reaction wrt reactant = power of molar conc of reactant in rate eqn {[A]0 : rate of reaction does not depend on A}
overall order of reaction: sum of powers of reactants in reaction eqn
rate const: a const that molar conc of reactants are multiplied to obtain rate of reaction [rate const: for particular reaction at const temp, varies w/ temp for exo/endothermic reactions, k: units vary depending on overall order]
Determination of order of reaction
(1)Initial rate method
rate = k[A]m[B]n
conc of A kept const, vary conc of B, rate of reaction measured
R1 = k[A1]m[B1]n
R2 = k[A1]m[B2]n
R1/R2 = {[B1]/[B2]}n
> conc of B const, vary conc of A
(2)Conc of reactant-time graph
zero-order reaction: straight line graph of constant -ve gradient (shows rate of reaction is independent of conc of reactant) [rate of reaction = grad of conc-time graph]
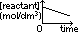
non-zero order reaction: curve

(3)Rate of reaction-reactant conc graph
zero order: straight line, zero grad
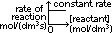
1st order:st line through origin, grad = constant
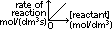
higher order: curve
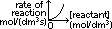
for nth order: st line through origin obtained if graph of rate reaction- (mol/dm3)n {rate of reaction of nth order ∝ [reactant]n}

(4)Half-life of reaction
half-life: time taken for conc of reactant to fall to half original value
if half-life = constant > 1st order
higher orders: not constant
Measuring rate of reaction (based on property of reactant / product)
(1)Polarimetric method (hydrolysis of sucrose) (for optically active subs)

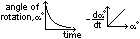
conc of sucrose ∝ angle of rotation of plane polarized light
use polarimeter to measure angle
conc of sucrose decreases > angle decreases
plot angle-time graph
plot gradient-angle graph > st line through origin > rate = k[sucrose]
(2)Colorimeter method (for subs w/ colour)
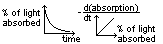
colour intensity ∝ conc of subs
colorimeter detects change in intensity (decreasing- reactants, increasing- products)
plot intensity-time graph > plot gradient-intensity graph
(3)Titrametric method (catalytic decomposition of H2O2)
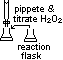
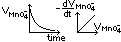
pipette H2O2 at diff times > titrate w/ KMnO4 > obtain [H2O2]
[H2O2] ∝ V(MnO4-) used
plot V(MnO4-)-time graph
plot gradient-V(MnO4-) graph > st line through origin > rate = k[H2O2]
(4)Volumetric method (catalytic decomposition of H2O2)
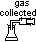
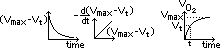
Vmax ∝ initial conc of H2O2
Vt ∝ conc of H2O2 used at time t
(Vmax - Vt) ∝ conc of H2O2 left at time t
plot (Vmax - Vt)-time graph
plot gradient-(Vmax - Vt) graph > st line through origin > rate = k[H2O2]
Collision theory of reaction
fro chem. reaction to occur, reactant particles must collide
not every collision causes a reaction
reaction occurs when energy of collision greater or equal to activation energy / energy barrier of reaction (activation energy, EA = min energy required to start a reaction by breaking bonds in reactant particles)
factors affecting reaction rate
conc of reactants, pressure (gaseous reactants), temperature, surface area (of solid), intensity of light (photochemical reactions), presence of catalyst
Concentration (rate constant not affected)
reactant conc increased > reaction rate increased
higher conc > collision freq increased > more collisions w/ energy getgetgetget EA > increase reaction rate
Pressure (rate constant not affected)
pV = nRT => p = nRT/V => p ∝ n/V (conc) [const temp] => pressure ∝ molar conc of gaseous products
higher pressure > higher conc > collision freq increased > more collisions w/ energy getgetgetget EA > increase reaction rate
Temperature (rate constant k changes w/ temp, rate const ∝ temp)
temp ∝ reaction rate
usually: small temp increase > large reaction rate increase
small temp rise: tremendous increase of no of molecules w/ energy equal or greater than EA according to Botlzmann distribution of molecular energy
Botlzmann distribution of molecular energy: shows distribution of molecular energies in a gas at const temp

no of molecules w/ energy E at temp T = Ae-(E/RT) [R = rate const, A = const ]
area under distribution curve = total no of gas molecules
curve only meets energy axis at infinity energy
only molecules w/ energy getgetgetget EA react on collision
temp increase > Boltzmann distribution curve shifts to right (higher energy) w/ lower peak
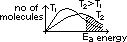
small temp increase > large increase in no of molecules w/ energy getgetgetget EA > more collisions per second w/ energy getgetgetget EA > increase reaction rate
Surface area
finer/smaller physical size of solid reactant > greater exposed area to fluid reactant > more effective collisions w/ energy getgetgetget EA > increase reaction rate
Intensity of light (for photochemical reactions)
intensity ∝ reaction rate
Catalyst
catalyst: subs that alters rate of reaction w/o being chemically changed at end of reaction
+ve catalyst: increase reaction rate by using different reaction mechanism in which EA lowered to EA`

collision freq & more molecules w/ energy getgetgetget EA` increases > reaction rate increased
-ve inhibitor: decrease reaction rate
catalysed reactions
Haber process: 3H2 + N2 <(Fe)> 2NH3
Contact process: 2SO2 + O2 <(V2O5)> 2SO3
manufacture of margarine: vegetable oil + H2 >(Ni)> margarine
enzymes: biological catalysts- hydrolysis of starch, replicatoin of DNA
Back to 'A' level notes index
Back to notes index
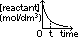 rate of reaction = -gradient of [reactant]-time graph
rate of reaction = -gradient of [reactant]-time graph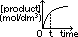 rate of reaction = -gradient of [product]-time graph
rate of reaction = -gradient of [product]-time graph