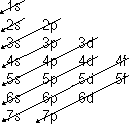ATOMIC STRUCTURE
Atoms: 3 fundamental particles-protons, neutrons, electrons(in energy shells = orbitals)
Other sub-atomic particles: positron, Quak particle, J-particle
| PARTICLE | REL. MASS | REL. CHARGE | SYMBOL
|
|---|
| Electron | 1/1840 (~0) | -1 | 0-1e
|
| Proton | 1 | +1 | 11p or 11H+
|
| neutron | 1 | 0 (electrically neutral) | 10n
|
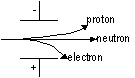
Deflection in electric field:
-neutrons; no charge > not deflected
-protons; +ve charge > -ve plate
-electrons; -ve charge > +ve plate (v.light > deflected alot)
Deflection in magnetic field:
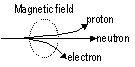
Deflection of some position: 2H+ & 4He2+
Nucleus of atom- contains (protons + neutrons) = nucleons, central mass of nucleus = nuclide (AZX)
A = mass #; total # of protons + neutrons in nucleus, numerically = to RAM (molar mass = RAM expressed in g/mol)
Z = atomic #; # of protons in atom
Isotopes = same element diff # of neutrons
Carbon: 12, 13, 14 (14C formed in outer atmosphere from 14N hit by fast-moving neutrons caused by cosmic rays: 14N + 1n > 14C + 1H)
Hydrogen: 1, 2,3 (2H/2D: deuterium, 3H/3T: tritium)
Isotopes: stable / unstable, unstable = radioisotopes, decay naturally > new element + radiation
Uses of isotopes
Medical (radiotherapy): kill cancer cells- 60Co (γ-rays v.strong), 32P/90Sr (weaker radiation > less penetrative- skin cancer)
Study of metabolic pathway: 32P in fertilizers- how plants absorb PO43-, 14C- photosynthesis (plants) & protein synthesis (animals)
Carbon-12 dating: ratio 14C: 12C > estimate age of wooden object; up to1000's of years only
Rock dating: ratio 238U: 206Pb > estimate age of Earth; (40-400) million years
Electron arrangement
Electrons- arranged in energy shells which are quantized
| Principal Quantum # | Energy shell
|
|---|
| 1 | K-shell (1st shell)
|
| 2 | L-shell
|
| 3 | M-shell
|
| 4 | N-shell
|
| 5 | O-shell
|
| 6 | P-shell
|
| 7 | Q-shell
|
| more elements > 8 | R-shell
|
In shells > subshells/orbits which have orbitals- regions in space (3D) where an e- most probably found
| Princiapl Q# | Energy shell | Subshells
|
|---|
| 1 | K | 1s
|
| 2 | L | 2s, 2p
|
| 3 | M | 3s,3p,3d
|
| 4 | N | 4s,4p,4d,4f
|
Each subshell contains a number of orbitals: s = 1, p = 3, d = 5 (dx2, d x2- y2, dxy, dxz, dyz), f = 7
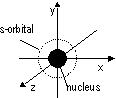
s-orbitals: spherical and symmetrical, p-orbitals: dumb-bell-shaped (3 sub-orbitals; px, py, pz)
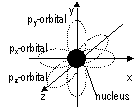
2nd Q#, l; corresponds to orbit:- l = 0:s,1:p, 2:d, 3:f
3rd Q#, ml; max # of orbitals in orbit
Rules of Electronic configuration of atoms
-each orbital contains max 2 electron of opp spin [(anti-)clockwise]
-for degenerates (orbitals of same energy) electrons fill every orbital w/ parallel spin before pairing up
-electrons fill orbital w/ lower energy 1st before ones w/ higher energy
Ground state: electronic config w/ lowest energy level
Order of filling orbitals
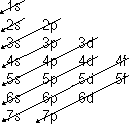
Chromium: [Ar] 3d54s1 (more stable energetically)
Copper: [Ar] 3d104s1 (more stable)
Same electronic config = isoelectronic
In periodic table: each group has same valence shell electronic config: Group VIII = ns2np6
3d, 4d & 5d elements = transition elements
4f & 5f elements = inner transition elements (man-made, radioactive) (4f- lanthanides, 5f- actinides)
Ionisation energy (IE)
Conditions: 1mole, gaseous atom, isolated (no inter-atomic attraction), valence electrons in ground state
1st ionisation energy of an element = min energy absorbed to remove 1 mol of valence e- (in ground state) from 1 mol of isolated gaseous atoms of the element to form 1 mol of isolated, gaseous, singly-charged ions
M(g) - e- >(ΔHIEI)> M+(g), ΔH= +ve
2nd IE = min energy absorbed to remove 1 mol of 2nd valence e- (in ground state) from 1 mol of isolated, gaseous uni-positive atoms of the element to form 1 mol of isolated, gaseous, doubly-charged ions
M+(g) - e- > ΔHIEII > M2+(g), ΔH= +ve
nth IE; M(n-1)+(g) - e- > Mn+(g), ΔH= +ve
Factors affecting magnitude of 1st IE
positive nuclear charge attraction: from protons in nucleus, greater atomic # > more protons > valence electron more attracted > greater IE
atomic radius: larger radius > less attraction of valence electron > smaller IE
across periods IE ^ (generally- more protons & smaller atomic radii > more attraction)
exceptions:- period 2: Be & N, period 3: Mg & P -due to extra stability w/ filled s config- Group II and ½ filled p config- Group V > more energy absorbed to remove e- [(½) filled orbitals > more stable]
down groups IE decreases: atomic radius ^ due to e- screening- repulsion bet e-s in shell > valence e-s far from nucleus > nuclear charge attraction decreases
transition metals:across periodic table- IE very little fluctuation
graph of lgIE vs # of e-s remove shows difference in successive IE's (pts before large gap= # of e-s in valence shell)
Back to 'A' level notes index
Back to notes index