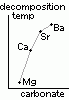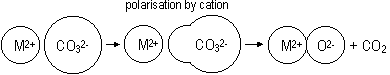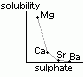| |
reaction w/ water |
product |
| Mg |
v.slow w/ water at room temp, more readily w/ steam Mg(s) + H2O(g) >(heat)> MgO(s) + H2(g) |
MgO- basic oxide, insoluble in water |
| Ca |
readily w/ cold water: Ca + H2O > Ca(OH)2(s) + H2(g) |
Ca(OH)2- basic hydroxide, slightly soluble in H2O |
| Sr |
readily w/ cold water: Sr + H2O > Sr(OH)2(aq) + H2(g) |
Sr(OH)2- basic hydroxide, soluble in water |
| Ba |
violently w/ cold water: Ba + H2O > Ba(OH)2(aq) + H2(g) |
Ba(OH)2- basic hydroxide, soluble in water |
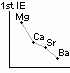
reactivity w/ water: Mg < Ca < Sr < Ba
| |
reaction w/ water |
product |
| MgO |
MgO(s) + H2O(l) > Mg(OH)2(s) Mg(OH)2(s)  Mg2+(aq) + 2OH-(aq)(low conc) Mg2+(aq) + 2OH-(aq)(low conc) |
MgO: sparingly soluble > weakly basic, pH ~8 |
| CaO |
vigorous + exothermic: CaO(s) + H2O(l) > Ca(OH)2(s) Ca(OH)2(s) + aq  Ca(OH)2(aq) Ca(OH)2(aq)  Ca2+(aq) + 2OH-(aq) Ca2+(aq) + 2OH-(aq) |
Ca(OH)2: slightly soluble > weakly alkaline sol called limewater |
| SrO |
vigorous + exothermic: SrO(s) + H2O(l) > Sr(OH)2(aq) Sr(OH)2(s) + aq  Sr(OH)2(aq) Sr(OH)2(aq)  Sr2+(aq) + 2OH-(aq) Sr2+(aq) + 2OH-(aq) |
Sr(OH)2: v.soluble > strong alkaline, pH ~13 |
| BaO |
vigorous + exothermic: BaO(s) + H2O(l) > Ba(OH)2(aq) Ba(OH)2(s) + aq  Ba(OH)2(aq) Ba(OH)2(aq)  Ba2+(aq) + 2OH-(aq) Ba2+(aq) + 2OH-(aq) |
Ba(OH)2: v.soluble >strong alkaline, pH ~13 |
CaO(s) = quicklime Mg2+(aq) + 2OH-(aq)(low conc)
Mg2+(aq) + 2OH-(aq)(low conc) Ca(OH)2(aq)
Ca(OH)2(aq)  Ca2+(aq) + 2OH-(aq)
Ca2+(aq) + 2OH-(aq)  Sr(OH)2(aq)
Sr(OH)2(aq)  Sr2+(aq) + 2OH-(aq)
Sr2+(aq) + 2OH-(aq) Ba(OH)2(aq)
Ba(OH)2(aq)  Ba2+(aq) + 2OH-(aq)
Ba2+(aq) + 2OH-(aq)
 Mg2+(aq) + 2OH-(aq)(low conc)
Mg2+(aq) + 2OH-(aq)(low conc) Ca(OH)2(aq)
Ca(OH)2(aq)  Ca2+(aq) + 2OH-(aq)
Ca2+(aq) + 2OH-(aq)  Sr(OH)2(aq)
Sr(OH)2(aq)  Sr2+(aq) + 2OH-(aq)
Sr2+(aq) + 2OH-(aq) Ba(OH)2(aq)
Ba(OH)2(aq)  Ba2+(aq) + 2OH-(aq)
Ba2+(aq) + 2OH-(aq)