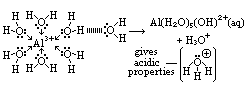
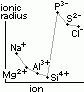
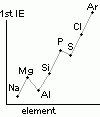
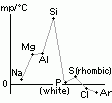
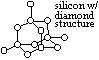
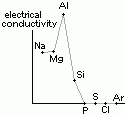
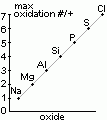
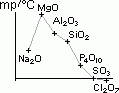
| oxide | Na2O | MgO | Al2O3 | SiO2 | P4O10 | SO3 | Cl2O7 |
| reaction w/ water | vigorous > NaOH | slow > Mg(OH)2 | insoluble | insoluble | reacts > H3PO4 | reacts > H2SO4 | reacts > HCl4O |
| equations | Na2O(s) + H2O(l) > 2Na+(aq) + 2OH-(aq) | MgO(s) + H2O(l) > Mg(OH)2(s), Mg(OH)2(s) > Mg(OH)2(aq) > Mg2+(aq) + 2OH-(aq) | P4O10 (s) + 6H2O(l) > H3PO4(aq) | SO3 (l) + H2O(l) > H2SO4(aq) | Cl2O7(l) (dichlorine heptoxide) + H2O(l) > 2HCl4O(aq) [chloric(VII)/ perchloric acid] | ||
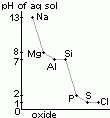
| pH of aq sol | 13 | 8 | 7 | 7 | 2 | 1 | 1 |
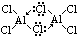
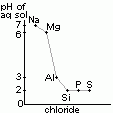
| chloride | NaCl | MgCl2 | Al2Cl6 | SiCl4 | PCl5 | S2Cl2 |
|---|---|---|---|---|---|---|
| reaction w/ water | solid > dissolves (no hydrolysis) | slightly hydrolysed > weakly acidic | covalent chlorides > hydrolysed > acidic sol [due to HCl(aq)] | |||
| pH of aq sol | 7 | 6.5 | 3 | 2 | 2 | 2 |
 Mg2+(aq) + 2Cl-(aq)
Mg2+(aq) + 2Cl-(aq) Al(H2O)5OH2+ + H+(aq)
Al(H2O)5OH2+ + H+(aq)