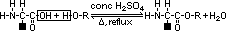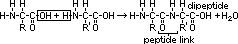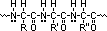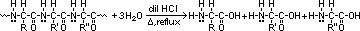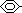 NH2 + HNO2 + HCl >(NaNO2/dil HCl, <5°C)>
NH2 + HNO2 + HCl >(NaNO2/dil HCl, <5°C)>  + 2H2O
+ 2H2O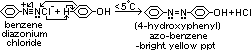
| TEST | phenylamine (aromatic) | phenylmethylamine (aliphatic) |
| + Br2(aq), room temp | white ppt |
no reaction |
| + NaNO2 & dil HCl at 5°C, + phenol | orange azo dye formed | bubbles of nitrogen gas, no reaction w/ phenol |
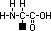 aminoalkanoic acid
aminoalkanoic acid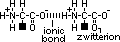
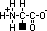
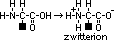
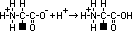
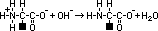
 ,
,