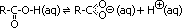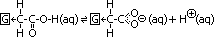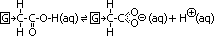ORGANIC ACID
MF: CnH2nO2 (alkanoic acid)
functional group: carboxylic group: -C(=O)OH (H attached to O can be ionised > H+/H3O+ > acidic properties)
Organic acids in nature
methanoic/ formic acid: in ants
ethanoic acid: in vinegar (vinegar: 7% ethanoic acid)
citric acid (white crystalline solid at rtp): fruits
lactic acid: in sour milk, produced in muscles during exercise when O2 lacking> cramps
benzoic acid (white solid at rtp): in some plants (sodium benzoate: C6H5C(=O)O-Na+ used as preservative)
steoric acid: CH3(CH2)16C(=O)OH
Formation of alkanoic acids
Lab preparation
Oxidation of 1° alcohols or aldehydes
reagent: acidified oxidising agent
condition: heat under reflux
R-CH2OH + [O] >(oxidising agent, heat, reflux)> R-C(=O)OH + H2O
R-C(=O)H + [O] >(oxidising agent, heat, reflux)> R-C(=O)OH
observation: excess alcohol / aldehyde: KMnO4: purple > colourless, K2Cr2O7: orange > green
Hydrolysis of alkanonitrile
reagent: dil HCl
condition: heat under reflux
R-CN + HCl + 2H2O >(heat, reflux)> R-C(=O)OH + NH4Cl [-CN hydrolysed to -COOH]
Hydrolysis of amide or acid amide
reagent: dil HCl
condition: heat under reflux
R-C(=O)-NH2 + HCl + 2H2O >(heat, reflux)> R-C(=O)OH + NH4Cl [-C(=O)NH2 hydrolysed to -COOH]
Hydrolysis of ester
reagent: dil HCl / dil H2SO4
condition: heat under reflux
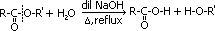
Industrial preparation of ethanoic acid
reagent: bacteria
condition: O2 available
CH3CH2OH + O2 >(bacteria)> CH3COOH + H2O
Physical properties
-lower members: liquids w/ sharp, characteristic smell of vinegar, higher members: white, waxy solids
-higher mp,bp than expected due to H-bond
-lower members: soluble in water, higher members: insoluble in water (H-bonds bet acid molecules too strong)
Acidic properties
-Neutralised by alkalis/ bases
reagent: NaOH(aq)
condition: room temp
R-C(=O)OH + NaOH > R-C(=O)O-Na+ + H2O
-Reaction w/ Na2CO3
reagent: Na2CO3 (aq)
condition: room temp
2R-C(=O)OH + Na2CO3 > 2R-C(=O)O-Na+ + CO2 + H2O
observation: -white Na2CO3 dissolves, -bubbles of CO2 evovled
Formation of acid derivatives
alkanoic acid; -OH group replaced to form acid derivatives
-OH replaced by:
-OR > R-C(=O)OR: ester
-Cl > R-C(=O)Cl: acid chloride
-NH2 > R-C(=O)NH2: amide
-OC(=O)R > R-C(=O)OC(=O)R: acid anhydride
Formation of esters
reagent: alcohol, conc H2SO4
condition: heat under reflux
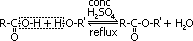
Formation of acid chloride
regaent: PCl5
condition: room temp
R-C(=O)OH + PCl5 > R-C(=O)Cl + POCl3 + HCl
observation: steamy white fumes of HCl (shows presence of -OH group)
Formation of amides
reagent: NH3 (aq)
condition: neutralise acid at room temp > ammonium alkanoate, NH4+ alkanoate: heat & dehydrate > amide
R-C(+O)OH(l) + NH3(aq) > R-C(=O)O-NH4+ >(heat-dehydrate)> R-C(=O)NH2(alkanoamide) + H2O
Reduction to 1° alcohol
reagent: LiAlH4 (tetrahydriodoaluminate(III)/ Lithium Aluminium Hydride) in dry ether
condition: ice cold (<10°C)
R-C(=O)OH + 4[H] >(LiAlH4, <10°C)> R-CH2OH + H2O
Acidity of organic acids
alkanoic acid molecule  alkanoate ion + hydrogen ion
alkanoate ion + hydrogen ion
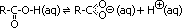
Structural factors influencing acidity
-Electron-withdrawing group attached to C chain
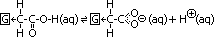
electron-withdrawing groups draw e-s from carboxyl group > reducing -ve charge density on alkanoate anion > stabilises anion & weakens O-H bond > more alkanoate ions and H+ formed > acidity increases
electron-withdrawing groups: halogen group, phenyl group; C6H5-, groups with unsaturated bonds (-C=O, -NO2)
-greater # of electron-withdrawing groups > alkanoate ion stablised more + O-H weakened more > stronger acid
-greater dist of electron-withdrawing groups from -C(=O)OH group > less e- withdrawing effect > weaker acid
-more electronegative electron-withdrawing group > stronger e- withdrawing effect > stronger acid
-Electron-donating group attached to C chain
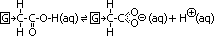
electron-donating group pushes e-s towards carboxyl group > increase +ve charge density of alkanoate ion > O-H bond stronger > less alkanoate & H+ ions > weaker acid
electron-donating groups: alkyl group; CnH2n+1-, hydroxy group; -OH , amino group; -NH2
-more electron-donating groups > more electron-donating effect > weaker acid
-greater dist from -C(=O)OH group > less electron-donating effect > stronger acid
Back to 'A' level notes index
Back to notes index
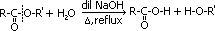
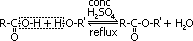
 alkanoate ion + hydrogen ion
alkanoate ion + hydrogen ion